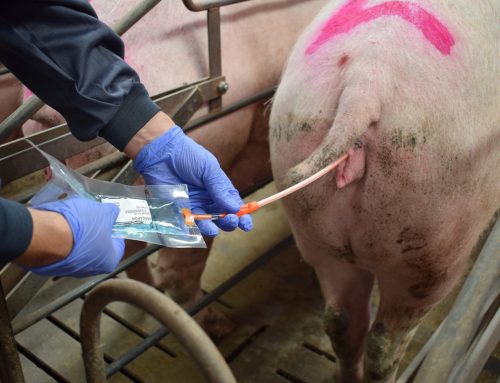
In this entry we will address some good laboratory practices to avoid mistakes during the quality evaluation of a boar ejaculate.
For all kind of analysis, we must standardize the lab methodology and also establish protocols. Therefore, regardless which operator carries out the analysis, the steps to follow are always going to be the same and there will be no variation in the results.
To begin with, we will expose generalities common to all systems.
Generalities:
- Homogenization of the sample in each stage (sampling, dilution and analysis): We must bear in mind that an ejaculate is not a homogeneous solution (seminal plasma, sperm, optional extender for the collection,…). In addition, many times for the analysis of an ejaculate, when some time passes after its extraction, a stratification of the components takes place and sperm cells deposits on the bottom of the container. Then, before taking any volume in each of the three stages, we must homogenize the sperm distribution to obtain a representative sample.
- Micropipetting: the handling of micropipettes can be a source of variation in the concentration evaluation. To avoid this, we must use them properly. To take the sample (ejaculate or the medium for dilution), we must press the plunger to the first stop and place the tip on the top of the liquid (depth 2-3 mm). Then, decrease the pressure of the plunger slowly to absorb the volume. If we released the plunger quickly, we could absorb air, taking an inaccurate volume, or even we could externally splash the tip. To expel the sample, we must lean the tip against the wall of the container (tube or eppendorf), and press the plunger to the first stop and then to the second slowly. We must prevent the tip from contacting the liquid, since if we have previously introduced it in the ejaculate, this could lead to variation..
- Verification and periodic equipment calibration: all equipment and instruments used in the laboratory must be checked and calibrated according to the manufacturer’s instructions. If after verification the equipment needs a calibration, it must be carried out (scales, micropipettes, colorimeters, etc.).
- Training of operators and subsequent follow-up: as we mentioned in the previous entry, no method of analysis is perfect as itself, as they need trained personnel in order to be properly operated. Therefore, whenever a new person is employed, he/she must be trained. Likewise, the workers´ good lab-practices should be reviewed periodically.
- All material contacting the ejaculate should be tempered at 37ºC to prevent from affecting motility parameters. Also the counting chambers and extender (when using CASA), the slides and covers (if subjectively evaluated with a microscope), the micropipette tips, the microscope slide, etc. must be kept at 37ºC through the use of equipment such as hot plates, dry bath for pipes, water bath, …
- Avoid the appearance of tertiary morphoanomalies. The primary and secondary ones appear during the formation and maturation of the spermatozoon. The tertiaries are due to an incorrect manipulation of the ejaculate by the operators. Generally, tails tend to appear curled when there are high temperature contrasts between the ejaculate and the material it contacts to. Also when a sample is diluted with a wrong saline dilution.
Next, we will also expose some peculiarities in each system.
Counting chamber for cells or hemocytometer (Bürker Chamber, Neubauer, Thoma, …)
Regarding the Bürker chamber (the one recommended by Magapor if this system is used), one should count the spermatozoa inside the small squares or in contact with 2 of the four edges that form the square (always follow the same criteria). We must do this for 40 squares in both grids (upper and lower), counting a total of 80 squares and obtaining an average data. In case there is a difference higher than 10% between both grids, the count should be repeated.
Colorimeter
- Switch it on some minutes before the first measurement, so that the light source gets stable.
- Avoid dirt in the buckets and do not touch the part where light strikes, since all this could influence the amount of light that will pass through the sample.
- As we mentioned in the previous entry, the calibration line of the colorimeter should be reviewed a couple of times a year.
CASA System
- Good focusing: When we notice that there is more than 5% of spermatozoa not detected during the analysis, it is because the image focusing is not correct. This will lead to underestimate the ejaculate concentration, in addition to not identifying the abnormal sperm cells correctly.
- As this type of chamber is filled by capillarity, we must ensure a complete filling of the counting chamber according to the exact volume indicated by the supplier. Besides, these counting chambers should be stored protected from dust and moisture, to avoid dirt or particles that may affect counting.
- Also, before starting to measure the different parameters, we must wait for the sample to stabilize. Thus, if we analyze just after filling the chamber, the movement of the sample can affect the evolution of the parameters.
- Take images or videos of the center of the counting chamber, and enough captures to get a correct representation of the ejaculate (it is usually indicated by the supplier).
We hope these tips may be useful to you. In following entries we will focus on flow cytometry.
Do not miss it !!!!